In a
New York Times article published in February 2002, James Glanz and Eric Lipton wrote:
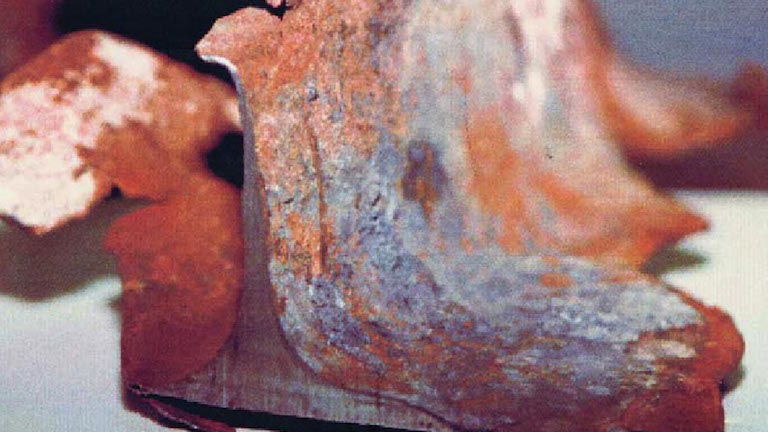
“Perhaps the deepest mystery uncovered in the investigation involves extremely thin bits of steel collected...from 7 World Trade Center.... The steel apparently melted away, but no fire in any of the buildings was believed to be hot enough to melt steel outright.... A preliminary analysis at Worcester Polytechnic Institute [WPI]...suggests that sulfur released during the fires—no one knows from where—may have combined with atoms in the steel to form compounds that melt at lower temperatures.” 1
The WPI professors, who were “shocked” by the “Swiss cheese appearance” 2 of the steel, reported their analysis in Appendix C of the
FEMA WTC Building Performance Study, making the following recommendation:
“The severe corrosion and subsequent erosion of Samples 1 and 2 are a very unusual event. No clear explanation for the source of the sulfur has been identified.... A detailed study into the mechanisms of this phenomenon is needed....” 3
A simple explanation for the source of sulfur, as well as the high-temperature corrosion and erosion, is “thermate,” which is produced when sulfur is added to thermite. In “
Revisiting 9/11—Applying the Scientific Method,” Dr. Steven Jones explains:
“When you put sulfur into thermite it makes the steel melt at a much lower temperature, so instead of melting at about 1,538°C it melts at approximately 988°C, and you get sulfidation and oxidation in the attacked steel....
“The thermate reaction proceeds rapidly and is in general faster than basic thermite in cutting through steel due to the presence of sulfur.” 4
 www.ae911truth.org
www.ae911truth.org